Call for Implementation of Biomarkers 2027

Problem
In the Netherlands, many biomarkers reach clinical validation but fail to achieve nationwide implementation. Fragmented infrastructure, limited reimbursement pathways, lack of harmonized guidelines, and insufficient coordination between stakeholders, delay or prevent integration into routine care. In addition, barriers in healthcare financing hinder sustainable uptake and scaling. As a result, access to biomarker-driven care varies across institutions, and patients do not consistently benefit from innovations that are ready for clinical use. Addressing these systemic barriers is essential to enable equitable access, effective scaling, and sustainable embedding of biomarkers in routine oncology care.
Ambition
The programme ‘Implementation of biomarkers’ aims to facilitate a fully validated biomarker into practice on a national scale. The programme is open for phase III studies with a clinical grade biomarker and/or for projects aiming at implementing clinical grade biomarkers into practice (TRL7-9). This goal should be achieved within the shortest, yet realistic timeframe possible. KWF funding will be adjusted or ceased when the biomarker reaches a stage where further development should be (partially) taken over by a (for profit) third-party.
Definitions
- Biomarkers: biomarkers are defined as objectively measurable biological or imaging-based characteristics that indicate cancer risk, abnormal or pathological processes, or responses to a (therapeutic) intervention. This includes molecular, genetic, proteomic, physiologic and imaging (e.g. histologic or radiographic) biomarkers used for early detection, diagnosis, prognosis, prediction of treatment response, or monitoring in oncology.
- Imaging biomarker: an imaging biomarker is a biological feature, or biomarker detectable in an image. Imaging is the technology; the biomarker is the standardized, reproducible measurement outcome obtained from imaging.
- Model-based biomarkers: biomarkers can be derived using analytical or computational models (e.g., statistical models from AI), provided that these models lead to a clearly defined, reproducible biomarker outcome that can be analytically and clinically validated. In imaging- and AI-based biomarkers, the biomarker is not the algorithm itself, but the standardized biomarker outcome (e.g., score, index, or classification) generated by the algorithm.
To ensure that the ambitious goals will be reached, but also to give room to adapt the project plan, a yearly evaluation and interview with the project team and its collaborators by an international evaluation committee will form an essential part of the programme. The evaluation committee will provide the project team with feedback and advice and can also give recommendations to adjust the project plan, budget or composition of the project team. It can also determine go/no-go moments and advise KWF to terminate the project prematurely if the ultimate goal is no longer deemed achievable (fail-fast principle).
To increase the likelihood of success, the project team will be asked to set up a separate expert panel (steering committee) that will provide support and advice on biomarker development. The composition of this panel needs to align with the needs of the specific development phase of the biomarker and may change over time.
To get first insights into medical, social or economic implications of the biomarker, a mini-Health Technology Assessment (mini-HTA) will be part of the evaluation. The mini-HTA will be performed for all full proposals by the same external partner to guarantee uniformity of the analysis. Contact with the external partner and payment of the mini-HTA will be arranged by KWF. In addition, for the runtime of the project, you will be required to have a HTA specialist in the team.
In scope
Projects in which a clinical grade biomarker will be qualified in an operational (clinical) environment (TRL 7 or beyond). This means that the performance, effectiveness, and reliability of the biomarker is evaluated under conditions that closely mimic everyday clinical practice, including workflow and equipment.
Important note: a discussion of the proposal with one of the biomarkers Science Liaisons at KWF is strongly recommended for submitting your proposal. This is to briefly discuss whether the proposal is a good fit for this programme (or whether it would fit better elsewhere) and what is required from you for the application. You can contact us at any time under [email protected]
Out of scope
- Biomarker projects below TRL7
- Measures without a direct biological or imaging-based link to cancer-related processes or treatment response.
- Biomarkers to identify/predict side effects of cancer treatment.
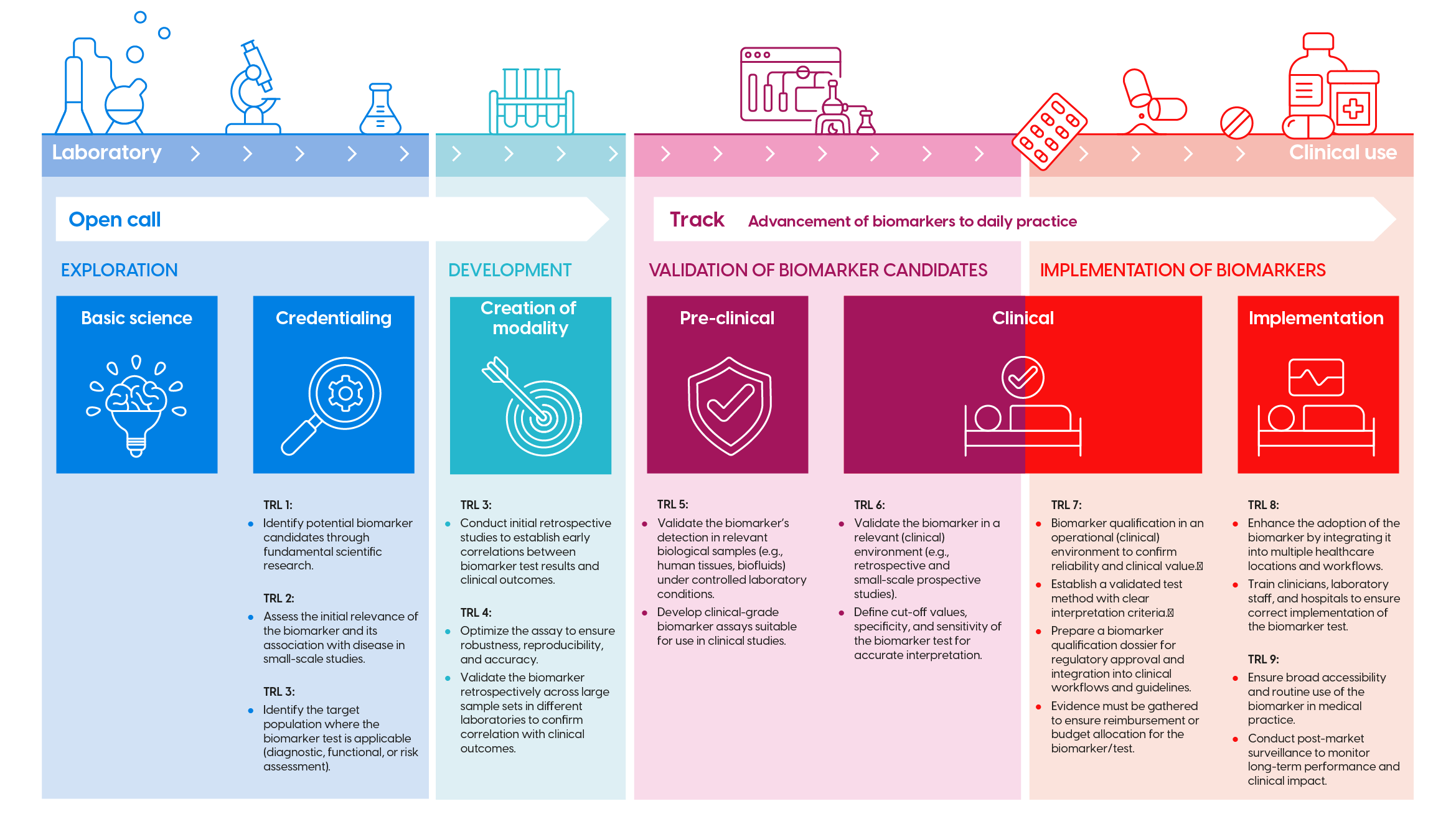
Biomarker development trajectory (click image to enlarge)
The development of biomarkers consists of successive research phases and Technology Readiness Levels (TRL). These are visualized in the infographic above (click to enlarge). For each phase, KWF has corresponding funding opportunities (open calls, theme call, etc.). Check the infographic to determine if your proposal fits the Programme Implementation of Biomarkers.
Terms & conditions
The KWF Financing Conditions 2025 and the KWF Accountants Protocol 2025 apply (see Downloads).
Specific guidelines on the process, characteristics and eligibility terms for the Implementation Call 2027 will be based on the current KWF Guidelines. Guidelines will include specific information on application requirements, preferences and recommendations, review procedure and timelines, and estimated total budget. Granted KWF projects will be funded under the current Funding Terms and Conditions.
Additional eligibility and requirements (in addition to the standard KWF funding conditions):
- Research type: (multidisciplinary) consortium addressing the required variety of disciplines with a minimum of 4 participating parties (which may include private partners)
- Consortium is supported by a project manager.
- Participating parties: all required expertise, including e.g., biostatistician, implementation science. Public/Private collaborations are encouraged if co-funding is in place. KWF holds the right to request additional information about the for-profit partner if this is deemed necessary.
- The patient perspective is structurally embedded throughout the project.
- Proposals explicitly address patient needs and include a description of the patient journey.
- Research phase: clinical/implementation.
- An HTA (incl. expertise in project team and sufficient budget) is a mandatory component of the application.
- Projects must address key conditions for sustainable implementation in routine care, including: integration into clinical guidelines and care pathways
- Before the start of the project, a steering committee approved by KWF must be established.
- Twice a year, a meeting will take place involving the project team, the steering committee, and KWF. The main task of the steering committee is to advise the project leader with the aim of maximizing the chance of successful implementation.
- Every year, the progress of the project will be evaluated by the evaluation committee. This includes whether milestones and go/no-go moments have been reached, but also project plans and whether the composition of the team fits the current development phase of the biomarker. The evaluation committee also evaluates the budget. A negative evaluation can lead to termination of the project. Therefore, it is strongly advised that no PhD candidate is assigned on the project.
In justified cases, applicants may deviate from specific eligibility conditions, provided that a clear and substantiated rationale is included in the application.
For-profit partners
KWF distinguishes between different roles for for-profit private partners, based on their responsibilities and contribution to the project. For-profit private partners can either be Private participating parties, Co-funders, or Service providers.
- A Private participating party carries substantial and financial responsibility for a part of the project, plus the dissemination and/or exploitation of the results. If a for-profit private party is responsible for the manufacturing, and/or has ownership of, or rights to, the biomarker test or product that is in development and provided in-kind, this party is also seen as participating party. Access to results/data and rights for commercialisation need to be formalized in a Collaboration Agreement and need to adhere to KWF’s standard Terms and Conditions. According to international guidelines, a participating for-profit private party can also have co-authorship in a publication if a scientific contribution has been made.
- A Co-funder provides financial and/or material contribution (in-kind and/or in-cash; e.g. a biomarker test or product that is already on the market) to the project but has no active involvement in the execution of the project. Access to project results (such as data) for a co-funder for commercial use shall be permitted only upon completion of the study, and under market conform conditions, which must be formalized in a co-funder agreement or material transfer agreement (MTA). A co-funder is not entitled to co-authorship, as the party does not provide a scientific contribution to the project.
- A Service provider is a (for-profit) partner that is involved in the project on a fee-for-service basis. Service providers have no obligations to own-contribution and have no rights to any results from the project. There is a maximum hourly rate for payroll costs for service providers which can be found in the KWF Tarievenbeleid 2026. The Project Leader is responsible for the inclusion of any VAT.
Apply for funding
The application procedure consists of a pre-proposal and full-proposal phase. Both should be submitted through our Grant Management System (KWF GMS) before their corresponding deadlines (see Timeline below).
PLEASE NOTE: Participating institutes not yet registered must submit a request through KWF-GMS at least 6 weeks prior to the call's closing date.
Timeline
| Opening pre-proposals: | 25 August 2026 |
| Closure pre-proposals: | 20 October 2026 (12.00 noon) |
| Opening full proposals: | 15 December 2026 |
| Closure full proposals: | 23 February 2027 (12.00 noon) |
| Interviews: | May 2027 |
| Funding decision: | June 2027 |
Recommendations and considerations
- Researchers are requested to contact the RIVM if their proposal involves any of the population screening programmes (BVOs), even if this is only expected in the future. If this has not yet been done, please send an email to [email protected].
- KWF is aware that there is more uncertainty when estimating the budget for activities that are far away in the future. Therefore, every year the budget will be re-evaluated and discussed with the evaluation committee. When preparing the budget, start with how you envisage the biomarker being used in clinical practice and then think of all the steps that need to be taken to get there. Do you already foresee a certain point in time where biomarker development is taken over by a for-profit partner?
Indicative budget and duration
Budget per proposal: 1-4 million
Duration: 3-5 years
Evaluation
A special international review committee (including patient perspective) will be selected for this call, consisting of experts in relevant areas. KWF uses three review criteria: relevance for KWF’s main goals, scientific quality and feasibility. At TRL 7–9, the focus of the evaluation is on demonstrated clinical utility, real-world impact, and sustainable nationwide implementation of the biomarker.
Interview
After evaluation of the full proposals the (multidisciplinary) consortia will be invited for an interview.