Call for Validation of biomarker candidates 2027

Biomarkers, as measured by (molecular) imaging, in blood, urine, stool or breath, are key to identify patients at risk of developing cancer, to predict/monitor progression or treatment responses and to detect recurrences.
Despite decades of discovery-driven biomarker studies, only a small number of biomarkers have been successfully validated and implemented in daily care. A reason is that translation and implementation of the biomarkers requires multidisciplinary teams that might differ in composition and expertise from the team making the initial biomarker discovery. Moreover, clinical decision-making often is based on the input of several biomarkers, again requiring a team with complementing expertises. Finally, it takes professional assay/software development, technical and clinical validation to set the stage for widespread clinical acceptance and use of new biomarkers.
Ambition
To clinically validate (combinations of) already known biomarkers that align with patient relevant outcomes or treatment responses and that can be used in daily clinical practice in the near future (TRL5/6). To achieve this, efforts are needed to set up collaborative, multidisciplinary studies with special attention to professional assay/software development and clinical translation. The focus of this call is to bring together the expertises in the field of biomarker research and development, the clinical care of end users and the needs of patients. By focusing on implementation rather than discovery, we strive to provide the appropriate framework and funding to realize the implementation of valuable biomarker candidate(s).
Definitions
- Biomarker
Biomarkers are defined as objectively measurable biological or imaging-based characteristics that indicate cancer risk, abnormal or pathological processes, or responses to a (therapeutic) intervention. This includes molecular, genetic, proteomic, physiologic and imaging (e.g. histologic or radiographic) biomarkers used for early detection, diagnosis, prognosis, prediction of treatment response, or monitoring in oncology.
- Imaging biomarker
An imaging biomarker is a biological feature, or biomarker detectable in an image. Imaging is the technology; the biomarker is the standardized, reproducible measurement outcome obtained from imaging.
- Model-based biomarkers
Biomarkers can be derived using analytical or computational models (e.g., statistical models from AI), provided that these models lead to a clearly defined, reproducible biomarker outcome that can be analytically and clinically validated. In imaging- and AI-based biomarkers, the biomarker is not the algorithm itself, but the standardized biomarker outcome (e.g., score, index, or classification) generated by the algorithm.
Requirements for AI-biomarker projects
Many AI-driven biomarker validation projects show strong technical potential but struggle to reach healthcare implementation due to challenges in data availability, clinical alignment, and scalability. To overcome these barriers, KWF co-funds the AI4HEALTH consortium, which adopts a system-level approach to AI in healthcare across the full AI lifecycle.
AI4HEALTH is a comprehensive national consortium that accelerates the development, evaluation, implementation, and scaling of safe, effective, and trustworthy AI in healthcare. AI4HEALTH is formed by public, private, policy and regulatory partners. It aligns multidisciplinary expertise across validation, data science, clinical practice, patient perspectives, implementation, ethics, and regulation.
For oncologic biomarker validation projects, AI4HEALTH supports project teams in strengthening study design, clinical relevance, validation strategies, data analysis, implementation, and scalability enabling faster and more sustainable translation into healthcare practice.
For all projects that include AI, engagement with the AI4Health consortium is mandatory. Applicants are expected to involve the consortium as a partner or advisor and to reflect this collaboration in their proposal.
If your project has an AI component, contact [email protected] as soon as possible so we can connect you to the AI4Health consortium.
In scope
Validation of already known biomarkers in the relevant environment that align with patient relevant outcomes or treatment prediction/ responses and that can be used in daily clinical practice to improve oncological outcomes in the near future (TRL5/6).
Out of scope
- Identification of biomarkers (TRL1-4)
- Measures without a direct biological or imaging-based link to cancer-related processes or treatment response.
- Biomarkers to identify/predict side effects of cancer treatment.
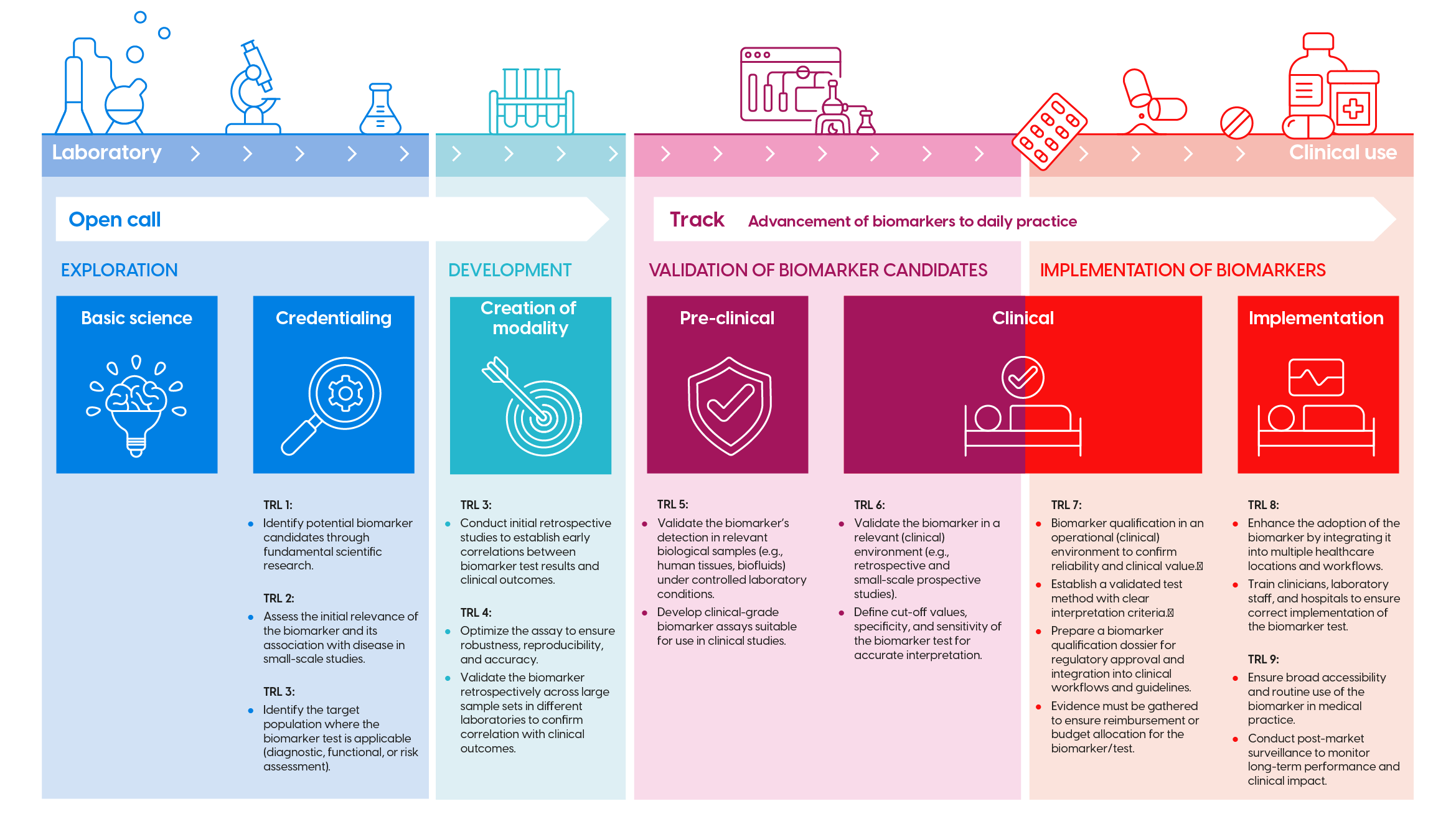
Biomarker development trajectory
The development of biomarkers consists of successive research phases and Technology Readiness Levels (TRL). These are visualized in the infographic above (click to enlarge). For each phase, KWF has corresponding funding opportunities (open calls, theme call, etc.). Check the infographic to determine if your proposal fits the appropriate call.
Terms & conditions
The KWF Funding Conditions 2025 and the KWF Accountants Protocol 2025 apply (see downloads).
Specific guidelines on the process, characteristics and eligibility terms will be based on the current KWF Guidelines. Guidelines will include specific information on application requirements, preferences and recommendations, review procedure and timelines, and estimated total budget. Granted KWF projects will be funded under the current Funding Terms and Conditions.
In addition, the following conditions apply:
- Research type: (multidisciplinary) consortium addressing the required variety of disciplines with a minimum of 4 participating parties (which may include private partners)
- Research phase: preclinical/clinical.
- Multidisciplinary team supported by a project manager.
- Participating parties: all required expertise, including e.g., biostatistician, HTA-expert (in-house service/not-for-profit organization). Public/Private collaborations are encouraged if co-funding is in place. KWF holds the right to request additional information about the for-profit partner if this is deemed necessary.
- For all projects that include AI, AI4Health is part of the consortium (participating or advisor)
- An (early)HTA (incl. expertise in project team and sufficient budget) is a mandatory component of the application.
- Applications must demonstrate robust statistical expertise, including active involvement in the project team and allocation of sufficient budget.
- The project addresses patient needs; including a description of the patient journey.
- The perspective of patients is incorporated. Patients/patient associations are closely involved during the set-up of the proposal, the conduction of the research, and the sharing of the (lay)results with the target group.
- Detailed developmental plan. Applicants are required to include a concise development plan describing how the biomarker will progress from clinical validation towards real-world implementation. This plan should clearly define the clinical and patient value it is expected to deliver (such as improved outcomes, reduced toxicity, or more efficient care), and the pathway to market/nationwide implementation. The development plan should explicitly address Health Technology Assessment (HTA) considerations, including anticipated evidence requirements for reimbursement and adoption, relevant comparators, and the expected impact on clinical practice and healthcare costs. Where applicable, regulatory strategy, stakeholder involvement (e.g. clinicians, patients, payers), and IP and commercialization strategy should be outlined.
In case you have a valid reason, you may deviate from the eligibility condition. This valid reason must be substantiated in the application.
For-profit partners
KWF distinguishes between different roles for for-profit private partners, based on their responsibilities and contribution to the project. For-profit private partners can either be Private participating parties, Co-funders, or Service providers.
- A Private participating party carries substantial and financial responsibility for a part of the project, plus the dissemination and/or exploitation of the results. If a for-profit private party is responsible for the manufacturing, and/or has ownership of, or rights to, the biomarker test or product that is in development and provided in-kind, this party is also seen as participating party. Access to results/data and rights for commercialisation need to be formalized in a Collaboration Agreement and need to adhere to KWF’s standard Terms and Conditions. According to international guidelines, a participating for-profit private party can also have co-authorship in a publication if a scientific contribution has been made.
- A Co-funder provides financial and/or material contribution (in-kind and/or in-cash; e.g. a biomarker test or product that is already on the market) to the project but has no active involvement in the execution of the project. Access to project results (such as data) for a co-funder for commercial use shall be permitted only upon completion of the study, and under market conform conditions, which must be formalized in a co-funder agreement or material transfer agreement (MTA). A co-funder is not entitled to co-authorship, as the party does not provide a scientific contribution to the project.
- A Service provider is a (for-profit) partner that is involved in the project on a fee-for-service basis. Service providers have no obligations to own-contribution and have no rights to any results from the project. There is a maximum hourly rate for payroll costs for service providers which can be found in the KWF Tarievenbeleid 2026. The Project Leader is responsible for the inclusion of any VAT.
Recommendations and considerations for applicants and reviewers
The Dutch Cancer Society includes a pre-proposal round for this call to be able provide recommendations for the researchers and input and/or guidance for the reviewing committee. Those additional considerations for proposals, which allow prioritization if necessary are:
- A proven regulatory/market-access strategy and/or regulatory support
- Make use of or participate in existing central facilities and (data) infrastructures (databases, biobanks, imaging data, etc.)
- If not yet participating, an advisory board with parties that are familiar with the implementation of a biomarker in daily practice is a pre.
- Researchers are requested to contact the RIVM if their proposal involves any of the population screening programmes (BVOs), even if this is only expected in the future. If this has not yet been done, please send an email to [email protected].
Indicative budget and duration
Budget per proposal: 0.8-2 million
Duration: 2-5 years
Apply for funding
The application procedure consists of a pre-proposal and full-proposal phase. Both should be submitted through our Grant Management System (KWF GMS) before their corresponding deadlines (see Timeline below).
PLEASE NOTE: Participating institutes not yet registered must submit a request through KWF-GMS at least 6 weeks prior to the call's closing date.
Timeline
| Opening pre-proposals: | 25 August 2026 |
| Closure pre-proposals: | 20 October 2026 (12.00 noon) |
| Opening full proposals: | 15 December 2026 |
| Closure full proposals: | 23 February 2027 (12.00 noon) |
| Interviews: | May 2027 |
| Funding decision: | June 2027 |
Evaluation
KWF uses three review criteria: relevance for KWF’s main goals, scientific quality and feasibility. At TRL 5–6, the focus is on robust clinical validation and preparing the biomarker for future implementation. Projects that include an AI component will be evaluated according to the same criteria and procedures as all other proposals.
A special review committee will be selected for this call, consisting of experts in relevant areas. Additionally, the full proposal will also be reviewed by the patient advocacy committee (PACO).
Patient Advisory Committee
The Patient Advisory Committee (PACO) plays a vital role in evaluating applications from the patient’s perspective. This committee ensures that the needs and experiences of patients are appropriately considered in project proposals.
To ensure that patient involvement is properly assessed and given due importance, the PACO review carries a fixed weighting. The PACO score will contribute 30% to the final score, while the (scientific) Review Board score will account for the remaining 70%. PACO members will base their assessment solely on the Dutch Summary and the Patient Participation section of your full proposal. Therefore, please ensure that these components are completed thoroughly and articulated clearly.
Interview
After evaluation of the full proposals the (multidisciplinary) consortia will be invited for an interview.